
It’s not easy putting this all together, ya know? Ctenodactylus gundi, or Common Gundi, is a soft-furred rodent that uses specialized bristles on their hindfeet for grooming. Image credit: vetchyKocour via DeviantArt.
Last week on Curious Sengi, we explored pectinate claws in birds, a structure that looks like an obvious comb-like grooming tool. However, as we have seen, the whole story is a bit more nuanced.
Our furry relatives have evolved a number of different strategies to clean and maintain their pelage, but here we are going to take a closer look at examples of mammalian comb-like structures, i.e., toe combs, tooth combs, and pectinate teeth.

Eastern Barred Bandicoot, Perameles gunnii. Image credit: Dave Watts via Arkive.
Some groups of marsupials are characterized by a curious fusion of toes in what is called a syndactylous digit, where the second and third digits of the hindfoot are reduced in size and bound together in the same sheath of tissue. Each digit remains distinct skeletally, but appear like a toe with two claws. Bandicoots, koalas, kangaroos, wombats, and the Australian possums have this syndactylous condition (Martin, Pine, & DeBlase 2001). Based on early observations of living marsupials, it was clear that the syndactylous digits were not rudimentary structures, but specialized grooming claws (Wood Jones 1925).

A stripey backside this magnificent deserves a proper grooming tool. Eastern Barred Bandicoot, Perameles gunnii. Image credit: Yale Peabody Museum / Curious Sengi.
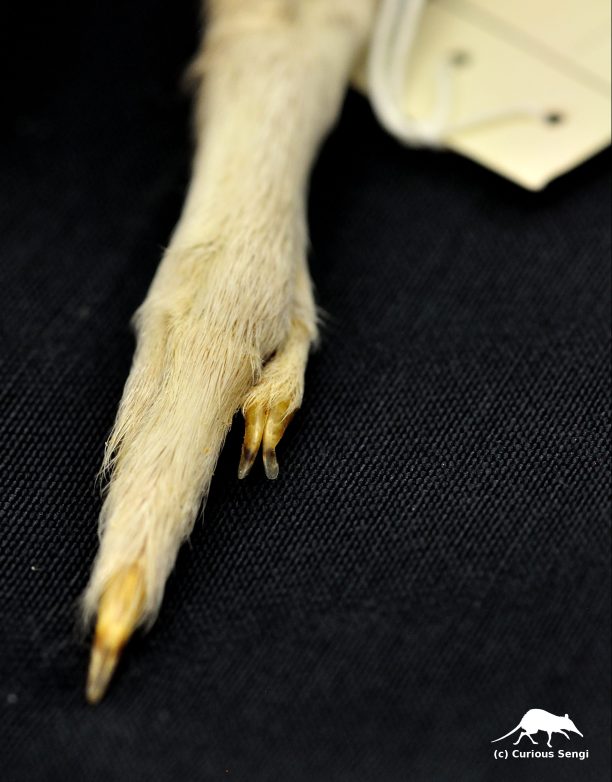
Detail of the second and third digit fused into a single syndactylous claw. Wood Jones (1925) declared that these are “. . . .definite hair combs, put to no other use whatever.” Image credit: Yale Peabody Museum / Curious Sengi.
Gundis (Ctenodactylus spp.) are highly social rodents found in the rocky desert areas of North Africa. They are appealingly plump and covered in soft, dense fur that provides both protective coloration and insulation from cold, high winds, and heat (Honig & Greven 2003). The two middle toes of the hindfoot — the second and third digits — are equipped with tufts of bristly hairs arranged in multiple rows. The lower combs, just above the claw, are short, strong, and curved. The upper combs, which are layered above the lower combs and merge into the fur of the leg, consist of longer and finer hairs (George 1978; Honig & Greven 2003).
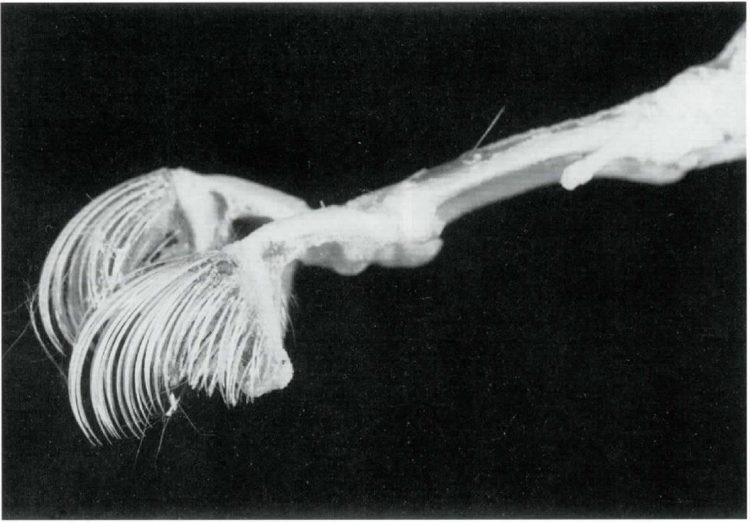
Skeleton of the left hindfoot with the second and third digits isolated to show the rigid hairs above the claws. The genus name, Ctenodactylus, aptly means “comb toe” (George 1978). Image credit: Honig and Greven 2003.
Given the importance of maintaining clean, ectoparasite-free fur in all mammals, why do gundis have a specialized grooming structure? George (1978) hypothesized that gundis have very sharp, curved claws necessary for scrambling about on rocks, but ill-suited for grooming soft, dense fur. Gundi fur also has a tendency to pull out easily, possibly as a defensive mechanism where quick shedding can facilitate slipping out of a predator’s grip. George filmed these animals grooming and saw that when scratching themselves, the claws were always turned under. This exposed the fur only to the toe combs and avoided contact with the sharp claws that could shred away hairs. Other very soft-furred rodents, such as chinchillas, also have toe combs.
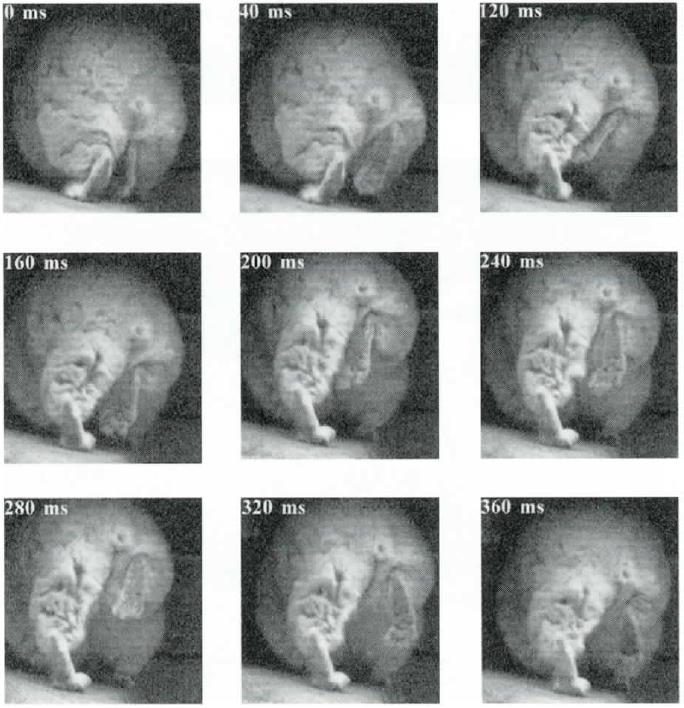
Gundis love scratching their rumps! George (1978) observed wild animals rump scratching six times an hour. This video sequence shows characteristic three-legged acrobatics involved in reaching the backside with the toe combs. Image credit: Honig and Greven 2003.

After each grooming session, the toe combs are carefully cleaned (Honig & Greven 2003). The bristles of the upper comb are clearly seen in this photograph. Image credit: cocoparisienne via Pixabay.
Moving from the foot end to the mouth end, many mammal lineages have independently evolved teeth in the lower jaw to act as combing structures. The most important example of this occurs in lemurs and lorises. The lower incisors and canines in these primates have each become slender, elongate, procumbent tines and then tightly packed together to form a combing array (Rose, Walker, & Jacobs 1981; Martin, Pine & DeBlase 2001).

Skull and lower jaw of the Diademed Sifaka, Propithecus diadema. The tooth comb appears as a tight grouping of procumbent incisors and canines at the very front of the mandible. Image credit: Yale Peabody Museum / Curious Sengi.
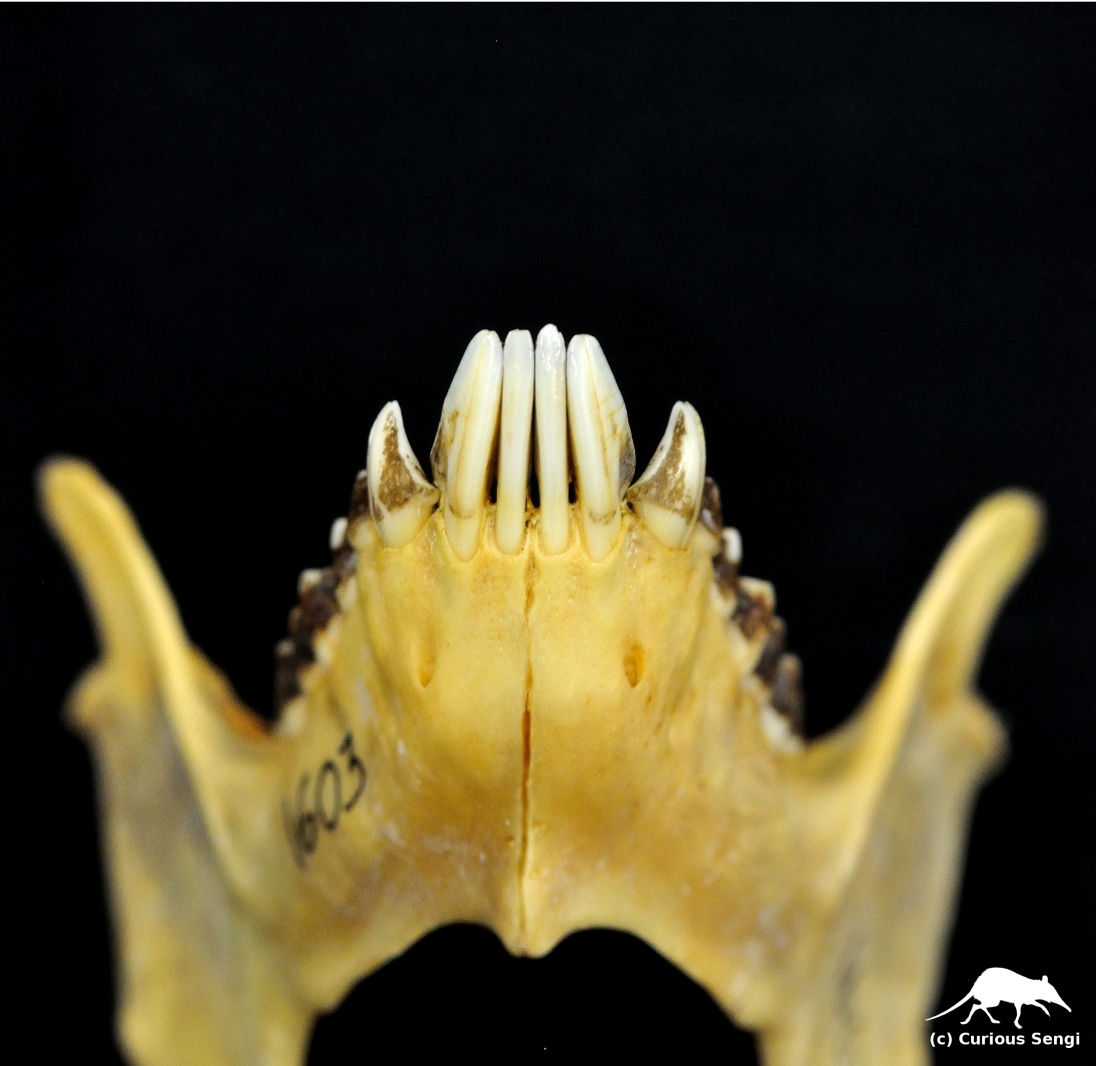
Unlike other lemurs, the sifaka’s tooth comb consists of only four teeth: two incisors and two canines (Martin, Pine, & DeBlase 2001). The sharp, canine-like fangs on either side of the tooth comb are actually premolars. Image credit: Yale Peabody Museum / Curious Sengi.
Tooth combs in primates has been a long part of their evolutionary history, with loris-like fossils from the Early Miocene (approximately 23 to 15 million years ago) showing tooth root positioning similar to the modern tooth comb array. The oldest definitive evidence comes from 7 to 8 million year old remains of Nycticeboides simpsoni from the Siwalik Group of Pakistan (Rose, Walker, & Jacobs 1981).
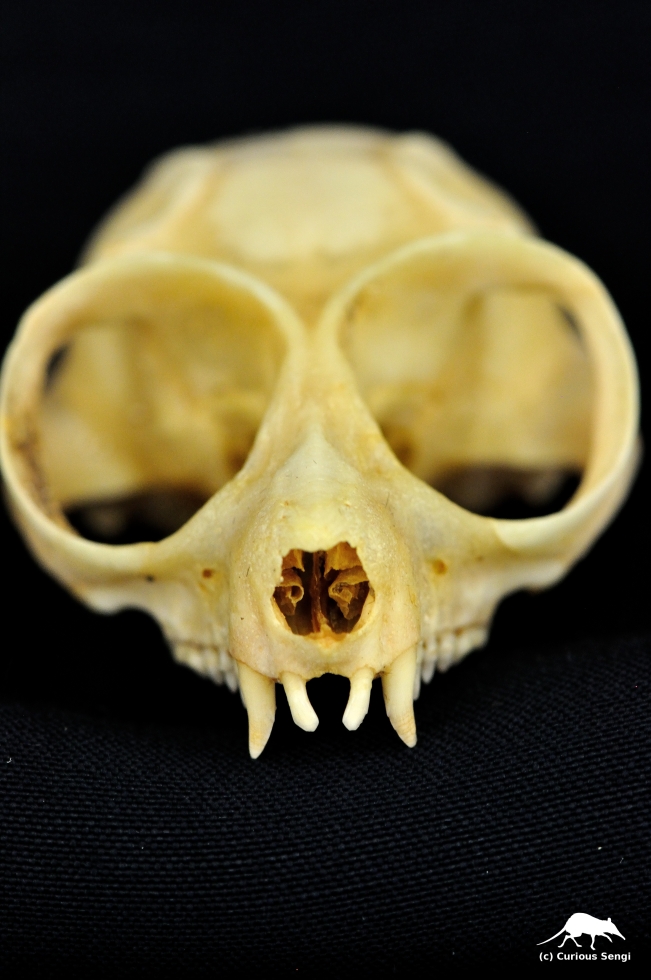
Skull of the Pygmy Slow Loris, Nycticebus pygmaeus. Note the upper incisors and canines are not tightly arranged into a combing structure. Tooth combs are limited to the lower jaws. Image credit: Yale Peabody Museum / Curious Sengi.
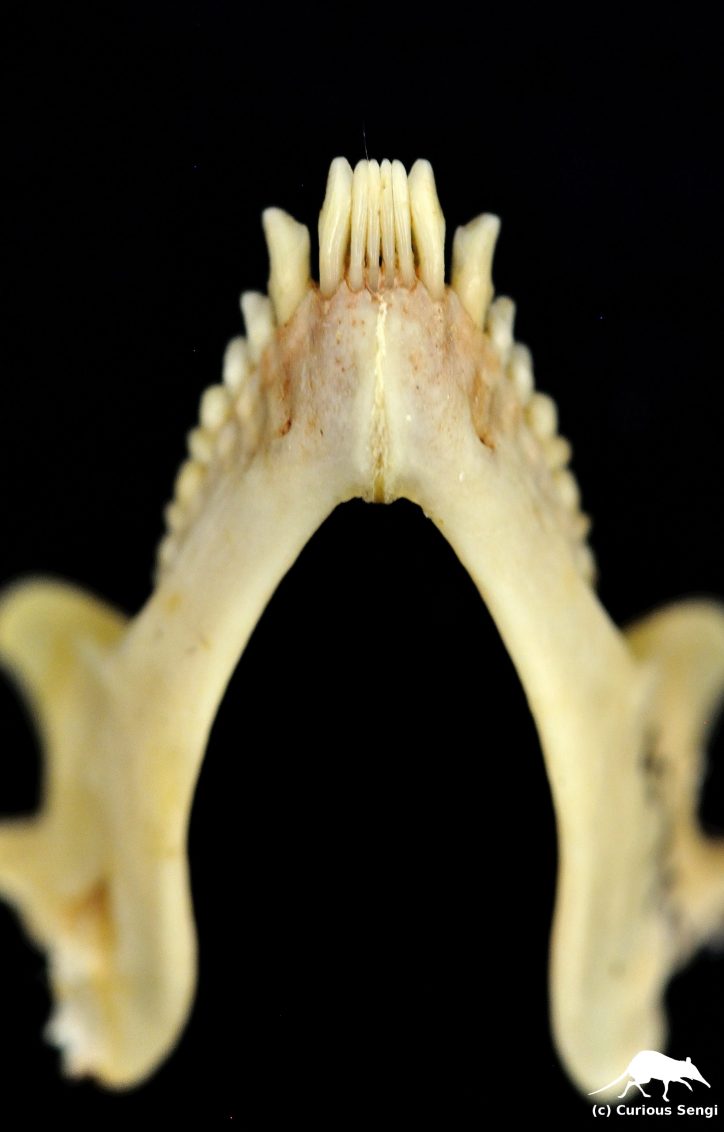
Loris tooth comb consisting of four incisors in the middle and two canines flanking the outside. Image credit: Yale Peabody Museum / Curious Sengi.
Another intriguing example of tooth combs in the fossil record emerges from the Early Eocene, approximately 56 to 41 million years ago. This comes from an enigmatic group of possibly carnivorous extinct mammals known as arctocyonid condylarths. With even their taxonomic affinity a big question mark, how was it possible to extrapolate grooming behavior from bones and teeth? Rose, Walker, and Jacobs (1981) used scanning electron microscopy to show distinctive microscopic wear patterns in known modern tooth comb groomers and compared them with the condylarths. A lifetime of combing through fur generates minute striations along the side edges of the teeth, where the hairs pulled through and left “whip marks.” The condylarths Chriacus sp. and cf. Thryptacodon sp from Wyoming both showed signs of these striations, indicating they were using their teeth as grooming tools.
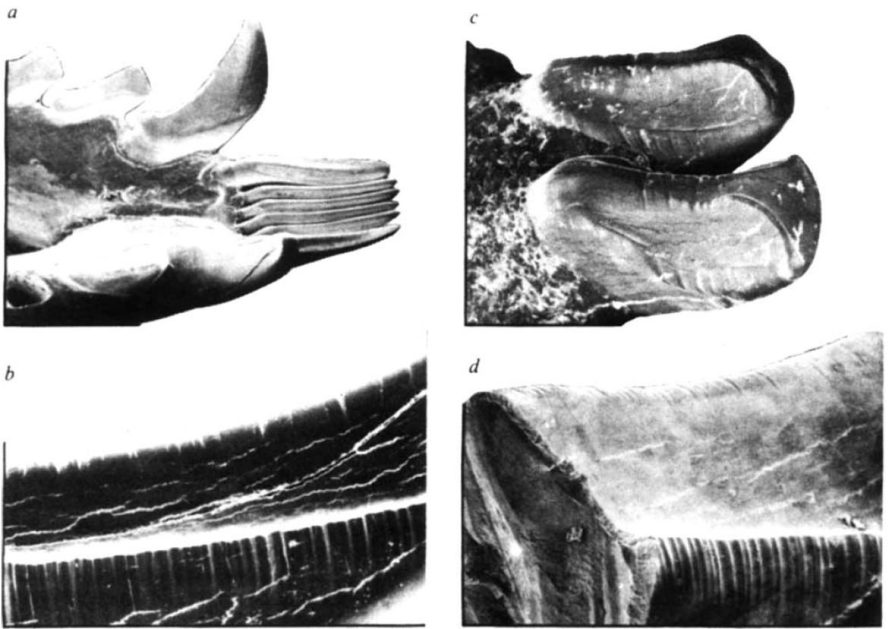
Scanning electron microscope (SEM) images of modern tooth combers on the left and condylarth fossils on the right. Both show signs of microstriations indicative of grooming. Image credit: Rose and Walker 1981.
Behavioral observations show primate and non-primate (e.g., colugos, tree shrews) tooth combs used in grooming, but also for collecting food, especially for gouging bark, scooping out fruit pulp, and scraping tree sap or gum. This draws up the question of whether tooth combs evolved first for grooming or for procuring food (Szalay & Seligsohn 1977; Rose, Walker, & Jacobs 1981). In 1972, Martin posits that food scraping is primary to grooming. Szalay and Seligsohn (1977) address this hypothesis, ultimately rejecting it upon functional grounds. Scraping bark or sap requires a strong, cutting edge. In generating this edge, it does not make functional sense to make the teeth longer and thinner, breaking up the cutting edge and making each tooth more susceptible to bending stress and potential breakage. In addition, in the wild forests bereft of dentists going on about flossing, long interdental spaces would become a nightmare of food impaction and rot. While the discussion continues about what tooth combs were originally adaptive for, there is little question that based on observations of live animals, the tooth comb is a multi-purpose tool.

Colugos, gliding mammals closely related to primates and tree shrews, possess pectinate teeth. In this tooth comb array, each incisor is divided into as many as 20 elongate tines (Aimi & Inagaki 1988). Lower jaw of the Phillipine “Flying Lemur”, Cynocephalus volans. Image credit: Yale Peabody Museum / Curious Sengi.
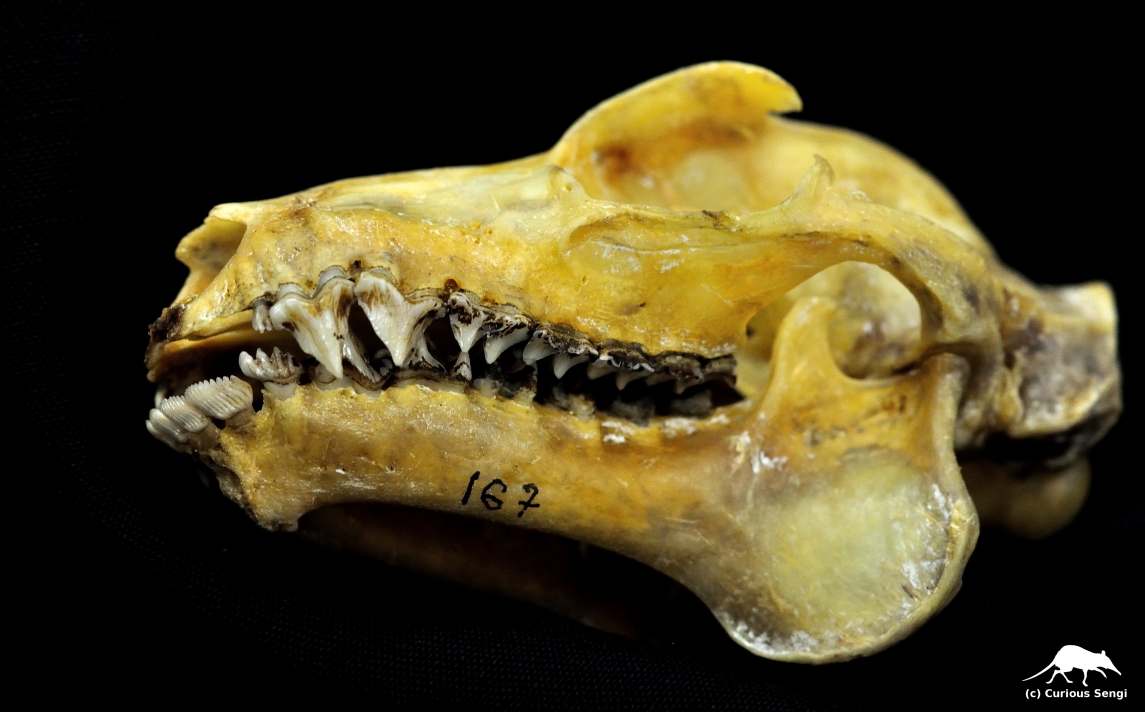
The pectinate teeth occlude against a toothless pad on the upper jaw. Colugos are noctural herbivores that forage on leaves, buds, fruits, and flowers. They have been observed using their pectinate incisors to scrape off the soft-tissue of tough leaves (Rose, Walker, & Jacobs 1981; Aimi & Inagaki 1988). Image credit: Yale Peabody Museum / Curious Sengi.

Mother colugo grooming her youngster. Image credit: Tumblr.
References
George, Wilma. 1978. “Combs, fur and coat care related to habitat in the Ctenodactylidae (Rodentia).” Zeitschrift für Säugetierkunde 43: 143 – 155.
Honigs, Sandra & Hartmut Greven. 2003. “Biology of the gundi, Ctenodactylus gundi (Rodentia: Ctenodactylidae), and its occurrence in Tunisia.” Kaupia 12: 43 -55.
Martin, Robert E., R.H. Pine, & A.F. DeBlase. 2001. A Manual of Mammalogy with Keys to Families of the World, 3rd ed. Long Grove, IL: Waveland Press, Inc.
Rose, Kenneth D. & Alan Walker. 1981. “Function of the mandibular tooth comb in living and extinct mammals.” Nature 289: 583 – 585.
Szalay, Frederick S. & Daniel Seligsohn. 1977. “Why Did the Strepsirhine Tooth Comb Evolve?” Folia primatologica 27: 75 – 82.
Wood Jones, Frederic. 1925. “The R.M. Johnston Memorial Lecture, 1925. The Mammalian Toilet and Some Considerations Arising From It.” Papers and Proceedings of the Royal Society of Tasmania: 14 – 62.
Pingback: Colugos, or Snapshot of Life in a Snuggie | Curious Sengi